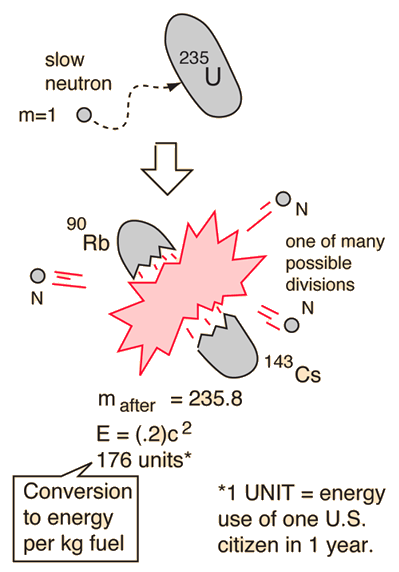
Suppose we hypothesize that the Ba and Kr isotopes in process (1) are the most common naturally found isotopes. The resulting 236U is very unstable, and instead of decaying via some already known form of radioactivity, it decays via fission. Since fission only occurs when neutrons are present, what really happens is that 235U first absorbs a neutron. For reasons to be discussed below, we will consider the example of fission of 235U. Natural uranium contains the two isotopes, 235U and 238U. This process was called fission, in analogy with the method of reproduction of one-celled animals. And krypton was not there: since it's a gas, it would have just drifted up into the air - not stayed in the uranium sample. Barium was identified via its chemical properties. Element number 36 is krypton, a rare gas. an alpha particle has Z = 2, thorium has Z = 90), here the uranium is split into two approximately equal parts: the second piece must have Z = 92 - 56 = 36. This suggested that a uranium nucleus has split into two parts but unlike normal radioactivity, where one part is very much smaller than the other (e.g. But the elements usually found in this way have high atomic numbers, close to that of uranium. Hahn and Strassman were chemists, and in the tradition of Marie Curie would not have been surprised to discover new elements produced by radioactivity (like thorium). In 1939 Otto Hahn and Fritz Strassman, experimenting with neutrons and a pure sample of uranium, found small traces of the element barium in the sample.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
